 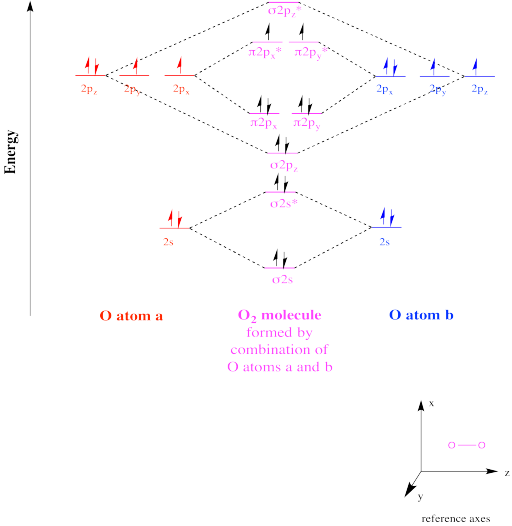
Energy levels for homonuclear diatomic molecules. We can match up the MO diagrams with the experimental data, such as bond length and bond strength, which suggests the bond order, and the magnetic properties, which tell us how many unpaired electrons the molecule has. Between N and O the σ(2p) crosses below the π MOs. (Also see that here.) The result is that sp-mixing decreases from left to right, because orbitals only mix well if they have similar energies. You can also see that the gap between the s and p-based orbitals gets bigger from left to right. The number of electrons increases as we move from right to left. The figure shows a summary of the energy levels, so we can see how they change. For instance, in a gas of Li metal, diatomic molecules will form. Note that the molecules that aren't common and stable might still be more stable in the gas phase than single atoms. Now we're ready to look at MO diagrams for the first row series. We would have to use complicated computer calculations to know exactly, but we can make pretty useful predictions just by thinking about it. The two in the middle might be harder to predict (especially because the molecule will use whatever ratios of s and p in the hybrids is best for lowering the total energy) but probably the lower one gets lower and the higher one gets higher. The 2p anti-bonding orbital becomes even more anti-bonding, and gets higher in energy. What effect does sp-mixing have on the MO diagram? The 2s bonding orbital becomes more bonding, and gets lower in energy. The slightly anti-bonding orbital is lower than the slightly bonding orbital, because it is still mostly made of s orbitals. The combinations are shown in the figure. Or you can think of it as making a bonding, 2 non-bonding, and an anti-bonding combination. We get a strongly bonding combination, a weakly bonding combination, a weakly anti-bonding combination, and a strongly anti-bonding combination. However, they are unequal hybrids, with more s in one and more p in the other.) We can picture sp hybrids making 4 combinations. (Basically they form sp hybrids, like you have seen before. And we can add some p-character to the s anti-bonding combination so that the overlap is worse, and it is less anti-bonding. This means that we can add some p-character to the bonding s combination, so the overlap is better. However, in elements with less difference between the 2s and 2p energies, like carbon, the 2s and 2p z orbitals can mix. Full valence MO diagram for F 2, showing all valence orbitals and electrons. You can see that including the 2s orbitals does not change the bond order, because both the bonding and anti-bonding combination are filled. So you get an MO diagram as shown in the figure. In molecules like F 2, they mostly interact separately, because the energy match between them is very bad. Do they do this separately? It turns out that sometimes they do and sometimes they don't. But we have to think more about the σ orbitals. The 2p orbitals that make π combinations don't do anything new. They are also smaller, so they have worse overlap with core orbitals on other atoms, and lower energy, so they have bad energy match with valence orbitals on other atoms. The core orbitals are completely full, so there can't be any net bonding between them. We don't have to worry about 1s orbitals, because their interactions don't have much effect on the properties of the molecule. Let's think about the orbitals we use to make MO diagrams for the first row elements, Li-Ne. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
